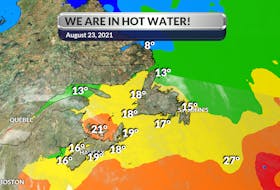
Some parts of our region have not received much rain this summer.
One day last week following a heavy downpour, the first in a good while, “Swiggy” tweeted this interesting question: “Just drove in the downpour, and no matter what road I was on, it looked like someone added soap. Kinda what you’d see at the car wash. What’s the phenomenon?”
No soap required for this car wash! The first two ingredients necessary are a liquid and a gas: in this case, water and air. So how does the air get trapped in the liquid to form bubbles? Asphalt is porous; the microstructure of asphalt consists of lots of tiny random open shafts throughout the otherwise solid material. Thanks to the capillary effect, water is sucked into those tiny tubes, and air is forced out. The air being forced out of the porous asphalt material and through the liquid on the surface creates bubbles.
Surface tension is the third ingredient - it controls the bubbles’ shape, size, and strength. Surface tension can be enhanced by surfactants: they are large molecules, which, when added to a liquid, increase the surface tension. The surface of a road accumulates lots of surfactants. In the summer, heat draws oil to the surface. Road oil that has come to the surface is sitting on top, mixing with rainwater.
It was very hot and dry and then it rained – bubbles for everyone!
- Read more Weather University columns.
- Have a weather question, photo or drawing to share with Cindy Day? Email [email protected]
Cindy Day is the chief meteorologist for SaltWire Network.